 
The valence shells of the inner transition elements consist of the ( n 2) f, the ( n 1) d, and the ns subshells. They are shown in green in Figure 8.3.6 8.3. The trends in ionisation energies across periods and down groups can be explained in terms of the atomic size, nuclear charge and the screening effect due to inner shell electrons. Inner transition elements are metallic elements in which the last electron added occupies an f orbital.C4.1b explain how observed simple properties of Groups 1, 7 and 0 depend on the outer shell of electrons of the atoms and predict properties from given trends down the groups.C4 Predicting and identifying reactions and products.C2.3.2 explain how observed simple properties of Groups 1, 7 and 0 depend on the outer shell of electrons of the atoms and predict properties from given trends down the groups Valence electrons are the electrons in the outermost shell, or energy level, of an atom.C2.3 How do metals and non-metals combine to form compounds?.6.13 Explain the relative reactivity of the halogens in terms of electronic configurations.6.5 Explain this pattern in reactivity in terms of electronic configurations. 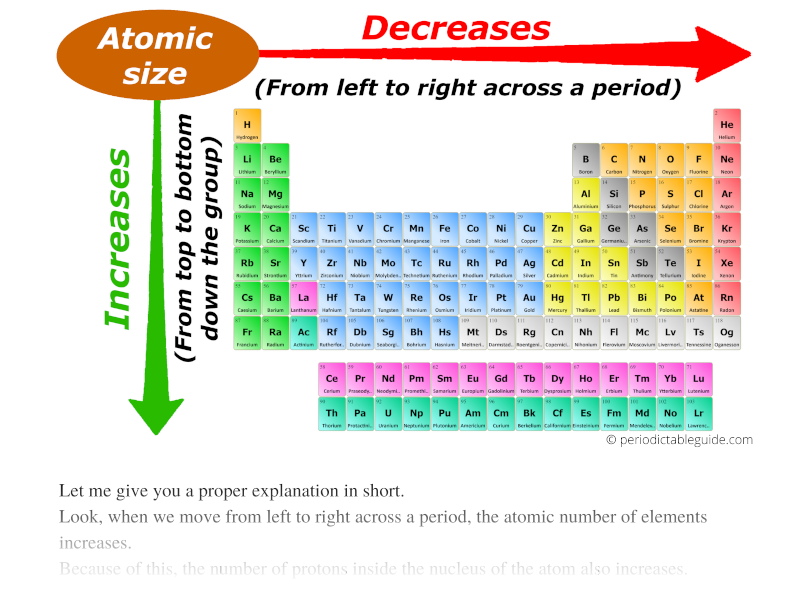
5.1 Atomic structure and the periodic table.Explain how the observed simple properties of Group 7 depend on the outer shell of electrons of the atoms and predict properties from given trends down the group. 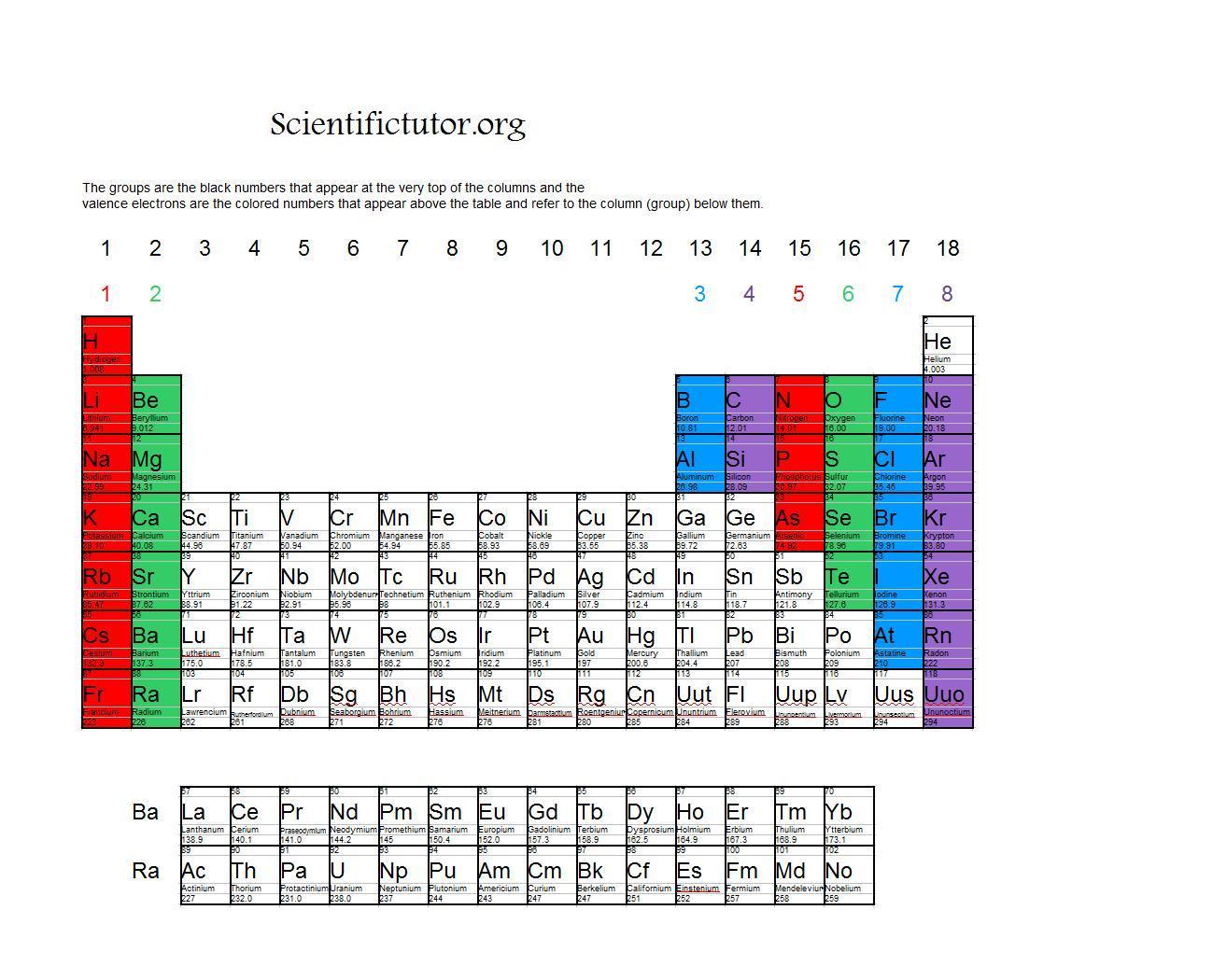
Explain how the observed simple properties of Group 1 depend on the outer shell of electrons of the atoms and predict properties from given trends down the group. Ionization energy: the amount of energy it takes to strip away the first valence electron Electronegativity: a measure of how tightly an atom holds onto its valence electrons Nuclear charge: the attractive force between the positive protons in the nucleus and the negative electrons in the energy levels.The more protons, the greater the nuclear charge. 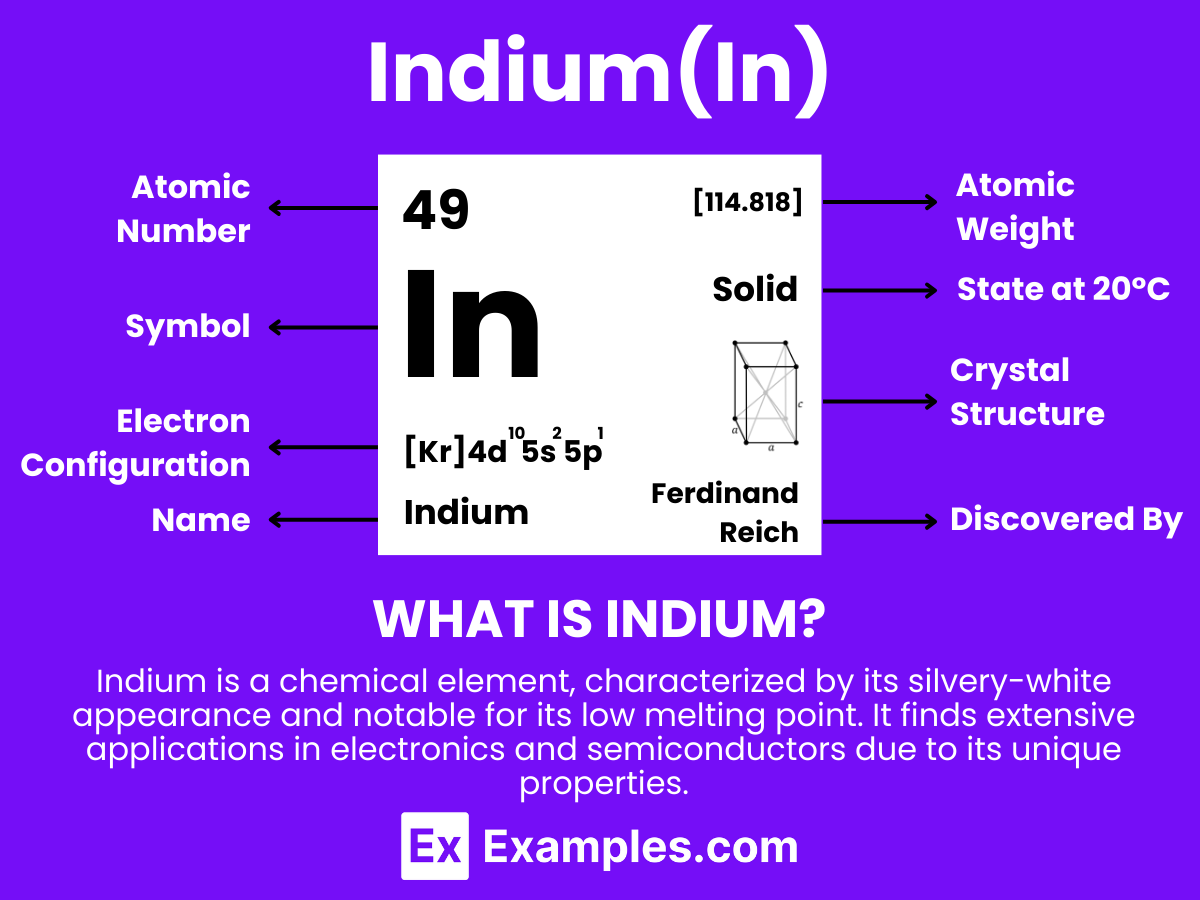
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
